Criteria for Choosing a Great Contract Research Organization (CRO)

Publication date: February 2024 / Download PDF Author: Viken Bartekian
Introduction
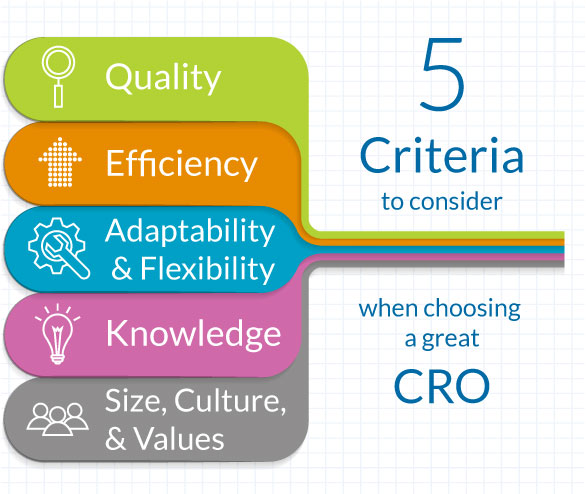
Outsourcing is often a necessity within the health care industry. Many pharmaceutical, biotechnology and medical device companies rely on outsourcing to Contract Research Organizations (CROs) as part of their strategy to advance to the next stage of clinical trials. As a matter of perspective, the CRO market size is expected to grow by USD 63.35 billion from 2023 to 2028. North America is estimated to contribute 40% to the growth of the global market during the forecast period.¹ Fueling this growth often places a burden on decision makers to make informed decisions regarding which CRO to outsource their clinical trial to. So, what makes a CRO stand apart? Simple, five important factors need to be considered: 1) Quality 2) Efficiency 3) Adaptability and Flexibility 4) Knowledge 5) Size, Culture and Values
You will notice that I have not mentioned the all-important cost factor. Understanding that for many companies, cost is an essential consideration to getting their clinical trial off the ground, we would also have to conclude that time is an important extension of cost. Just as the saying “time is money”, the clinical trial process has many moving parts, and all those parts are based on one constant factor, which is time! The lowest cost can be an influential factor to choosing a CRO. However, it’s important to understand that many CROs also use this as a bait tactic, only to generate change orders post contract win because they didn’t plan or estimate the time needed adequately.
Quality
The word quality is defined as the standard of something as measured against other things of a similar kind, or the degree of excellence of something.
To quote William A. Foster, “Quality is never an accident. It is always the result of high intention, sincere effort, intelligent direction, and skillful execution.”
We like rephrasing this quote into “Quality is an attribute we should always strive to achieve”. For this reason, quality always remains at the forefront of every clinical trial that Vantage BioTrials manages. It is embedded in our systems, procedures, and policies; in the way we train, monitor, and manage clinical trials. As experts in our field, we continuously implement risk mitigation by using Quality by Design (QbD) methodologies (to read more about QbD, refer to our previous Whitepaper entitled “Quality by Design in Clinical Research”).2
Having a quality mindset isn’t only to concentrate on results that matter. It’s to ensure that all results matter.
Efficiency
The term efficiency can be defined as the ability to achieve an end goal with little to no waste, effort, or energy. Being efficient means you can achieve your results by putting the resources you have in the best way possible. Put simply, something is efficient if nothing is wasted, and all processes are optimized. In other words, you will have mastered efficiency when you’re using the least amount of input to get the highest amount of output.
As a CRO, identifying efficiencies early on is crucial for the success of any clinical trial. But how early is too early? To give you an example, Vantage BioTrials applies Risk-based Quality Management (RBQM) strategies during the initial Request for Proposal (RFP) stage. Thoroughly reviewing a protocol and challenging a Sponsor is part of our process which helps align realistic expectations. This in turn presents an opportunity to explore changes required in order to achieve the most successful outcome for a project.
In our experience, open & transparent discussions help build a solid foundation towards maintaining an efficient long-term partnership.
Agile management is all about being adaptable and flexible in your approach, and it can be especially useful in the context of managing and monitoring of clinical trials. As the pioneering software engineer, Barry Boehm once said, “Agile methods derive much of their agility by relying on the tacit knowledge embodied in the team, rather than writing down their knowledge in plans.” It also helps to break down large projects into smaller, more manageable tasks, so that teams can easily adjust to changes and challenges as they arise. An agile approach helps keep timelines on track and ensures that everyone remains focused on the study’s ultimate goals.
Adaptability & Flexibility
Change is a constant factor in everyone’s life. Humans are often averse to change; we like a routine. Adapting to constant change is sometimes a challenge during the best of times. We can only imagine, and some of us have even lived it, changing ourselves or our environment thorough the worst of times.
As a CRO, with increased competition and a shifting regulatory landscape, we are constantly faced with significant challenges that push us to find ways to differentiate our service offering and remain competitive. As a specialized CRO, Vantage BioTrials is able to quickly pivot and apply tools and technologies that continue to conform with regulatory requirements. We remain vigilant, make quick decisions, and ensure little or no disruption to our services.
How quickly do we adapt you may ask? During the COVID-19 pandemic, we shifted from on-site monitoring to remote monitoring overnight. Even after some of our employees were poached to larger competitors, we quickly on-boarded local consultants with which we’ve formed strong relationships over the years. We went from accepting on-site Sponsor audits to remote ones by adopting a Quality Management System software. We adopted and onboarded a robust CTMS system within a month to track real time metrics of any given project.
Similarly, continuing to remain flexible is vital. Granted that in certain cases, flexibility becomes challenging. For example, a client wants your CRA to go monitor on-site across the country at the 11th hour, knowing that there are circumstances that would not allow you to do so (e.g. snow storms, ongoing airport strikes, etc.). Flexibility does not mean you jump when asked to. Flexibility is the willingness to change or compromise. Therefore, being solutions oriented is a key driver to remaining flexible. If one cannot physically go and monitor, then suggest additional remote monitoring as an option until on-site monitoring is possible again. If required, provide a valid explanation in defense of your decision. Remember that compromise swings both ways.
Financial flexibility is also important and often a driving force in the decision-making process of start-up biotechnology and medical device companies. A pricing model that Vantage BioTrials has utilized repeatedly is the fixed fee model:
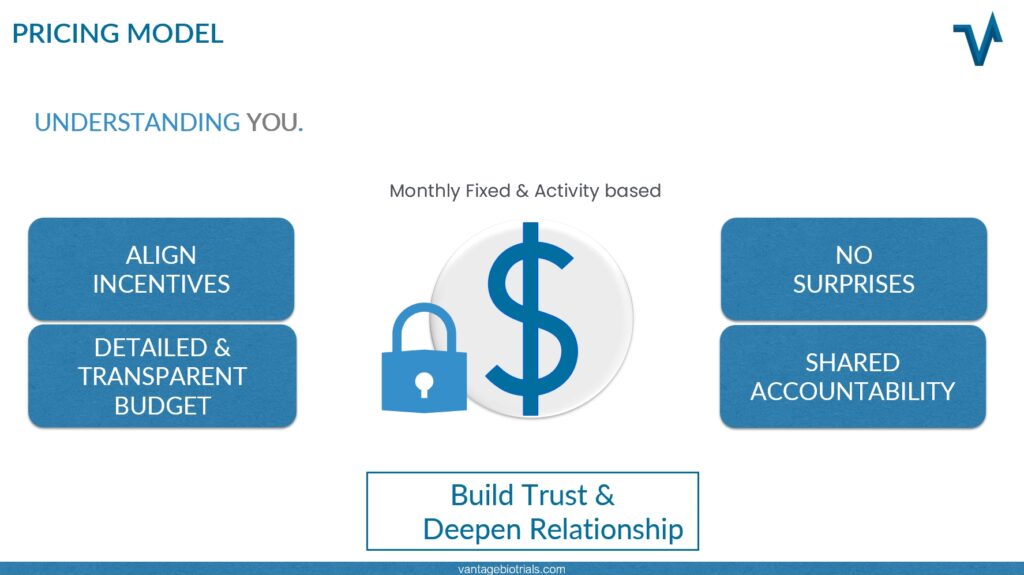
Not withstanding any amendments to the study design or protocol, this pricing model guarantees a set budget for the entirety of a project. It allows the tracking of projected vs. actualized amounts on a monthly basis. The benefits of this type of model includes:
- Aligning incentives
- Transparent budget visibility
- No unexpected fee or charges
- Shared accountability on commitments
- Builds trust and deepens relationships
Lastly, it is important to remain focused in providing operational excellence throughout the management of clinical trials. With our proven track record, Vantage BioTrials has managed clinical trials in various therapeutic areas from the most complex (rare disease, oncology, pediatric, respiratory, infectious disease) to simple (consumer health) and has repeatedly demonstrated its ability to adapt and achieve all clinical trial milestones.
Knowledge
Knowledge is a fundamental criterion when it comes to any business. It isn’t regarding how much you know, but also how much you can educate others with your knowledge.
With over 19 worldwide regulators (Health Canada, FDA, EMA, ANVISA, TGA, etc.) and a countless number of guidelines and standards within these governing bodies, the healthcare industry is one of the most heavily regulated. The different types of knowledge bases become even more crucial; regulatory, medical/scientific, quality standards (data, monitoring, biostatistics, risk analysis), operational best practices, budgeting, and technical. Demonstrating this knowledge base through examples of personal experiences, expertise and case studies is an important element that can strengthen the qualities of a CRO. Maintaining a sound training program/matrix ensures your organization’s ability to perform clinical trials which can in-turn instill the Sponsor’s confidence in your ability to delivery quality results.
Size, Culture and Values
The size of a CRO can be misleading when shopping for one. In fact, small size CROs such as Vantage BioTrials have proven time and again that we can manage clinical trials as good, if not better, than the larger CROs. Don’t look further than our achievement in 2021 when we announced the successful management and completion of a Phase IV observational, multicentre, postmarketing study in adult patients with a clinical diagnosis of moderate-to-severe Hidradenitis suppurativa (HS), approximately 12 months in advance.3
In the history of clinical trials, no other CRO (large or small) has ever come close to achieving this type of milestone. The result of which includes many of the factors listed above, plus, one important factor: our pharmaceutical Sponsor decided to take a chance on us, not due to cost, but because of our lean business model, which presents a more intimate, closer relationship with their team. This in turn leads to faster execution, easier conversations, and a less robotic and more genuine experience that generates trust.
Trust is the most important criteria when looking for a CRO as Sponsors need to learn to “let go” from their project and trust their CRO just as you would trust the electrician you hired to rewire your home. This is mainly the reason why having too much specialization in one domain can be counter-productive to the relationship and speed of execution. Developing trust and allowing your CRO to deliver results on their own terms also allows for mistakes to happen, for everyone to learn and grow, and for the relationship to flourish into a long-term partnership. Clinical trials are run by humans. To err is human, and mistakes will happen, it’s inevitable. Allowing your peers the space to prove their worth fosters deep appreciation for one another.
Respectively, it is the CRO’s duty to communicate properly, give regular updates, share enthusiasm, as well as to be fully transparent and accountable when there are red flags and concerns, as communication is key to the overall success of the project. The values of both companies need to be mutual and shared, and the Sponsor must fully explain the overview of the project, delegate tasks accordingly and the CRO needs to make the Sponsor comfortable by creating solid plans of action showcasing their full engagement to the project.
Having a vision explained, shared, and understood, as well as having aligned values, increases the chances of having matching company cultures and strong long-term harmonious relationships.
Final Thoughts
As competition grows, some CROs continue to over promise their capabilities and underestimate (or low ball) their costs. Sponsors are faced with challenging decisions on who to outsource to or sometimes they maintain the status quo and avoid changing CROs altogether because it’s perceptively too much of a burden. Factors other than cost need to be considered when deciding who to partner with.
As large CROs continue to remain siloed, it is important to choose a CRO that continues to offer flexibility in its service offering and fits your purpose. The same level of service should be given regardless if you’re a small biotech, medical device, or a large pharmaceutical company.
Building a strong relationship of trust will allow shared vision, values, and culture, which in turn maximizes chances of study success – always maintaining a clear Vantage Point!
About Vantage BioTrials
Vantage BioTrials is a leading Canadian Contract Research Organization (CRO) with a proven track record of successfully completing & delivering clinical trial milestones on time and on budget. Since our inception in 2007, we have used innovative clinical trial management strategies for the life science industry with a focus on patient safety & advancing new therapies to market. We offer pharmaceutical, biotech & medical device companies a complete and integrated set of full-service clinical trial management solutions, based on the principles of Quality by Design and Risk-based clinical trial management.
Contact us today at [email protected] and discover how we can add value to your next clinical trial program.
References:
2 https://vantagebiotrials.com/quality-by-design-in-clinical-research/


