Quality by Design in Clinical Research

Publication date: May 2015
Originally published in Journal of Clinical Research Best Practices, Vol. 11, No. 5, May 2015
Author: Vatche Bartekian
“Quality is never an accident. It is always the result of high intention, sincere effort, intelligent direction, and skillful execution.” – William A. Foster
In clinical research, “high quality” means that a study generates good, useful data, does not harm the study participants, and proceeds smoothly with few interruptions by errors or omissions. However, too many clinical research studies get mired in a time-consuming quality control (QC) process that generates numerous site monitor findings (e.g., missing data, ineligible study participants, other protocol deviations), data queries, and other after-the-fact indications that a quality problem has been found and needs to be fixed. A research site may be unable to deliver acceptable results, no matter how much the sponsor tries to help. In severe cases, the design of a study incorporates problems that make it impossible to achieve high quality results, no matter how well the study is conducted.
QC is just one part of a quality assurance (QA) program that ensures high-quality results can be achieved, with minimal quality problems that are quickly addressed. It is generally more efficient to build quality into a clinical study than to find and fix quality problems after the fact. This QA philosophy is called “Quality by Design” (QbD). The “Design” in QbD has two meanings: First QbD is a way to design high-quality processes. Second, QbD designs (intends) to improve quality. Both definitions are essential. As a metaphor, QbD means that instead of paving roads so traffic moves faster, don’t drive down the roads at all. Instead of creating more efficient inspection processes, eliminate the need for inspection. The fastest journey is no journey at all.
QbD does not mean designing error-proof processes, since clinical research is far too complicated and unpredictable to anticipate every possible problem in every possible circumstance. QbD, therefore, also means creating a process for identifying the quality problems that will occur throughout the life of a study and improving processes to minimize future problems.
Quality management is intertwined with risk management, since risk management is largely the practice of avoiding and mitigating quality problems. In other words, QbD not only builds quality in, it also builds risks out. For example, the risk of completing a study late can be greatly reduced by eliminating the potential quality problems that will slow it down.
Is turning a QC-focused QA process on its head worth the trouble? It is certainly possible to invest so much time in quality systems, including the QbD process itself, that the net payout is negative. However, organizations that have sensibly applied QbD have seen substantial positive payouts by minimizing quality problems, detecting those that occur sooner, reducing the cost of inspections, fixing problematic processes more quickly, and better understanding the quality dynamics of their clinical studies. In other words, with QbD, your trials can be faster, better and cheaper, not just better.
Implementation
It is hard to disagree with the concept of QbD, but implementation requires a change in mentality from conventional QC thinking, from the VP of Research to the clinical research associate or study coordinator.
A proven methodology for implementing QbD principles is the Plan-Do-Check-Act (PDCA) cycle. PDCA also supports continuous process improvement. The description of PDCA below uses a simple example to illustrate the cycle: Study sponsors often authorize data managers to fix certain obvious mistakes in the data generated by research sites. For example, if a case report form page lists concomitant medications taken by a study participant but the box for “Are there any medications this cycle?” is blank, the data manager may tick the box, thereby avoiding the time and cost of verifying the obvious correction with the site. These fixes are called “self-evident corrections” (SECs).
- Plan. Establish your quality objectives and define the necessary processes to achieve them. To start the planning process, identify opportunities that can yield the most value at the least cost. When possible, begin on a small scale, e.g., with a pilot project to confirm your assessment of the opportunity. Or, break a big problem into smaller, more manageable parts. You can utilize “Cause & Effect Diagrams” or the “5-Why’s” technique. For our example of SECs, begin by asking, for example, how many SECs are expected? How much time could they save? Are their definitions based on a coherent set of principles, e.g., are they allowed for endpoint data?
- Do. Implement the plan, execute the process, collect the data, and measure the results. Metrics are essential. For example, how many SECs are there for each site and each type of data?
- Check. Compare the metrics against your quality objectives. Use charts to identify trends, anomalies and problems with the processes themselves. Determine root causes. For example, was the SEC used wherever appropriate? If not, are the SEC definitions unclear? Is it easier for a data manager to send a query to the site than make an SEC?
- Act. This step is better defined as “Adjust,” since the actions usually consist of incremental adjustments to the goals, processes, metrics, etc. These adjustments can be implemented with a Corrective Action Preventive Action (CAPA) process. For example, clarify the SEC definitions. Automate the process of identifying possible SECs. Modify the incentives for making SECs vs. generating data queries. Perhaps raise the quality objective. Add a new metric: SECs/queries. When a process step proves its merit, make it a standard.
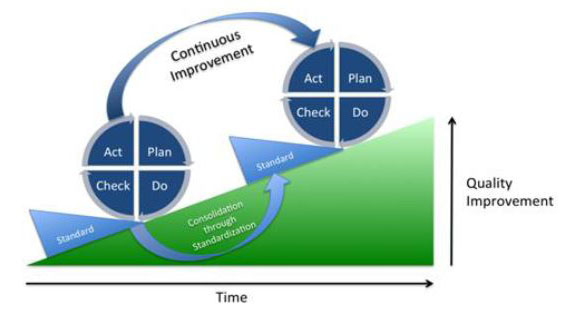
PCDA is a cyclical process. In other words, continue running it for the entire life of a study. The figure below demonstrates how continuous quality improvements can be achieved with the PDCA cycle over time:
About the Author
Author Vatche Bartekian is President of Vantage BioTrials, Inc., a global CRO based in Montreal, Canada.


